 
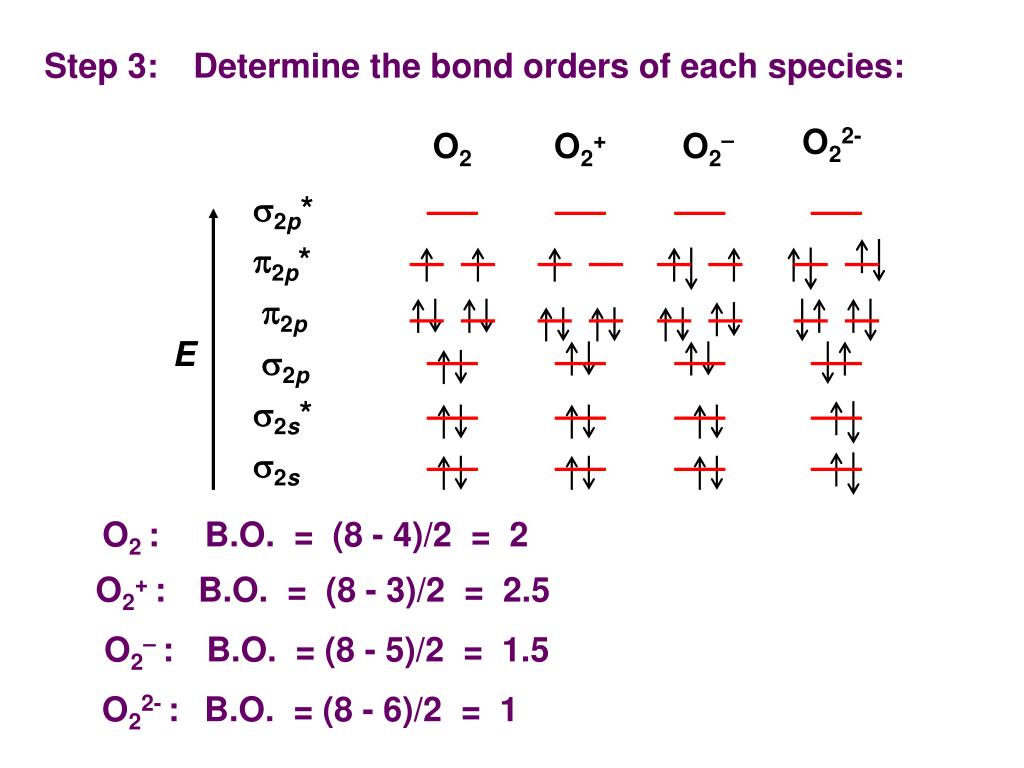
Molecular Orbital (MO) Diagram for O2 (2-) chemistNATE. So, without further ado, let us take a look at the. Molecular Orbital Theory - Bonding & Antibonding MO - Bond Order. Notice that all of the above are diatomic molecules. If you want #"O"_2^(+)#, take one electron out of the #pi_(2p_y)^"*"# orbital.\( \newcommand\): Hand-drawn molecular orbital diagram for dioxygen. Here is the list of MO diagrams shown in this gallery: O2 molecular orbital diagram. Lastly, #"N"_2# would have the #sigma_(2p_z)# above the #pi_(2p_x)# and #pi_(2p_y)#, whereas #"O"_2# would have it below. Two #2p_z# orbitals combine to give a #sigma_(2p_z)# bonding and #sigma_(2p_z)^"*"# antibonding MO.Draw the orbital diagram of 2040Ca+2 ion and state the number of three fundamental. These are the same energy as the #pi_(2p_x)# counterparts. (2s)2( 2s)2(2pz)2(2px)2(2py)2( 2px)1 Your first answer option is for N 2. Atoms of an element combine in small numbers to form molecules. Two #2p_y# orbitals combine to give a #pi_(2p_y)# bonding and #pi_(2p_y)^"*"# antibonding orbital.These are the same energy as the #pi_(2p_y)# counterparts. Two #2p_x# orbitals combine to give a #pi_(2p_x)# bonding and #pi_(2p_x)^"*"# antibonding orbital.(a) CN (b) CO (c) BeB (d) BC+ Figure 10.47 Molecular orbital diagram for nitric oxide (NO). Write the molecular orbital electron configuration of each, indicating the bond order and the number of unpaired electrons. 
The atomic orbitals of the O atoms overlap to form the and orbitals of the O2 molecule as shown in the diagram above. The correct explanation comes from Molecular Orbital theory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
